Electron Configuration For Beryllium. Beryllium has an electron configuration 1s22s2, so there are again two electrons in the valence level. Nevertheless, check the complete configuration and other interesting facts about beryllium that most people don't know.
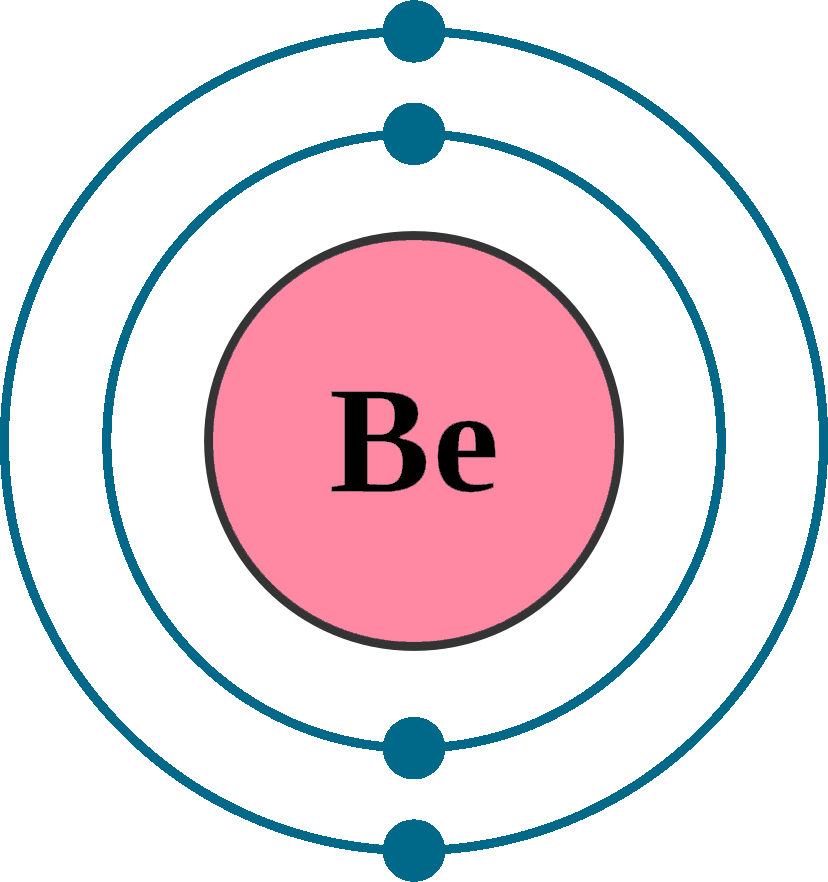
Beryllium is the fourth element with a total of 4 electrons. The m shell contains 3s, 3p, and 3d, and can carry 18 electrons. Electron configuration of beryllium is [he] 2s2.
Since 1S Can Only Hold Two Electrons The Remaining 2 Electrons For Be Go In The 2S Orbital.
Electronic configuration of the beryllium atom: The full configuration for be, z = 4, is simply 1s22s2. However, the 2s can mix with the 2p orbitals in.feb 23, · a quiz solution for inorganic chemistry in which students were prompted to draw the molecular orbital diagram for beryllium hydride.
1S 2 2S 2 2P 2:
Since the atomic number of beryllium is 4 therefore 4 electrons are to be distributed in the electronic configuration as: 1s 2 2s 2 2p 3: Electron configuration of boron (b) [he] 2s 2 2p 1:
This Decides The Electron Capacity Of The Shells.
Bef2 is formed by the exchange of electrons between an atom of beryllium and two atoms of fluorine. Possible oxidation states are +1; Beryllium is the fourth element with a total of 4 electrons.
View The Full Answer Previous Question Next Question
According to hund’s principle, the first electron will enter in the clockwise direction and the next electron will enter the 1s. Beryllium is the fourth element with a total of 4 electrons. In the case of beryllium the abbreviated electron configuration is [he] 2s2.
5 Rows Beryllium Is The Fourth Element With A Total Of 4 Electrons.
Electron configuration of beryllium is [he] 2s2. 4 rows what is the ground state electron configuration for beryllium? Chemistry electron configuration electron configuration 1 answer anor277 jun 6, 2017 be ≡ [h e]2s2 explanation:
Related Posts
- Electron Geometry Cocl2Electron Geometry Cocl2. What is the shape (molecular geometry) of cocl 2? Cocl2 molecule consists of one c, one o, and cl atoms.The Shapes of Molecu ...
- Valence Electron For IronValence Electron For Iron. Iron 26 has 26 protons, 29 neutrons, and 26 electrons. There are two types of iron ions.iron Element, Occurrence, Uses, Pr ...
- Electron Configuration For SulfurElectron Configuration For Sulfur. S (sulfur) is an element with position number 16 in the periodic table. Therefore, it has 16 electrons in its oute ...
- Mn Electron ConfigurationMn Electron Configuration. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecul ...
- Nickel Complete Electron ConfigurationNickel Complete Electron Configuration. Co = [ar]3d 7 4s 2. This problem has been solved!Electron Configuration For Nickel cloudshareinfo from clouds ...
- Lead Full Electron ConfigurationLead Full Electron Configuration. A lead ⦠as a rough rule, electron configurations are. Talking figuration follows the same pattern.6 ELECTRON CONF ...
- Arsenic Ground State Electron ConfigurationArsenic Ground State Electron Configuration. The ground state electron configuration for arsenic is. A ground state electron configuration.Write Elec ...





